TG and RAS – Holy Grail in cancer drug development
RAS proteins have potential to become oncogenic if any of the three encoding genes (HRAS, KRAS and NRAS) mutate. RAS as a cancer target has gained a rather notorious reputation over the last four decades, primarily because of its widespread prevalence among different cancers, which makes it an obvious target to pursue. However, so far there are no RAS-targeted treatment options approved. As Ledford (2015) summarised, RAS is a “high hanging fruit” in the oncology research arena. Since RAS mutations are present only inside malignant cells and represent well characterised neoantigens, the logic is that an RAS vaccine could induce cancer-specific, T-cell-based immunity without harming healthy tissues. The intracellular location of RAS proteins makes it impossible to use targeting molecules that recognise cell surface targets (eg antibodies). In contrast, T-cells recognise intracellular antigens as peptide fragments of those when presented in complex with MHC molecules on the target cells, thus making the RAS mutations visible to RAS specific T-cells generated by TG vaccination.
RAS mutations that turn normal cells into cancerous cells are single base alterations meaning that only one amino acid substitution turns the expressed protein oncogenic. In total several different amino acid substitutions are found and usually only one is present in a tumour. TG01 consists of seven peptides that mimic the most common position 12 and 13 RAS mutations in pancreatic and colorectal cancer, while TG02 consists of eight peptides covering the most common position 12 and 13 mutations of RAS in cancer in general, including colorectal and lung cancer.
With currently available tools, stratifying patients according to what RAS mutations are present is hardly feasible on a large scale. Therefore, the TG platform inventors decided to produce a vaccine that would cover most of known RAS mutations. Weden et al, the original research team, which included Targovax’s co-founder and current Chief Technology Innovation Officer Jon Amund Eriksen, published long-term (10+ years) follow-up data from the early clinical trials conducted by Norsk Hydro. Resected pancreatic patients were treated with TG01 as a monotherapy (no chemotherapy after the surgery), either with a single TG peptide (nine patients) or TG01 (11 patients) (Exhibit 6). The findings were that Targovax’s vaccine induced long-lasting T-cell responses. Analysis showed median survival of 28 months, with 20% of the 20 patients still alive 10 years post-surgery. While this was a retrospective analysis with no control arm, for comparison, the reported historical median survival in similar group of patients with standard of care is 20.0 months with 7.7% 10-year survival.
Exhibit 6: 10-year survival of resected pancreatic cancer patients, monotherapy with TG
|

|
|
|
In contrast to ONCOS-102, TG vaccines are administered by intradermal injection also with the immune booster GM-CSF. From the perspective of the cancer immune cycle, once injected TG peptides are picked up by antigen presenting cells, which correspond to a step later than ONCOS-102 (step 2 in Exhibit 2). Nevertheless, the TG vaccine is still an immune primer since the goal is to activate tumour-specific T-cell production, therefore like with ONCOS-102, this mechanism of action lends to a combination treatment with CPIs. The anticancer effect of TG is comprised of T-cells produced due to direct TG stimulation, but once cancer cells are attacked and killed, they release more cancer antigens, which further stimulate specific anticancer immunity.
Fresh data from the main group in the Phase I/II TG01-01 study
The ongoing Phase I/II trial with resected pancreatic cancer patients is the most advanced trial with the TG platform so far. Exhibit 7 shows the trial design. In total, 32 patients have been enrolled into two cohorts. TG01 was administered immediately after the surgery and there was no control arm. In order to establish the influence of chemotherapy timing, patients were given different dosing regimens.
Exhibit 7: TG01-01 study design
|

|
|
|
Targovax presented the results from the first cohort (n=19) in June 2017 at ASCO. Of the 19 patients enrolled, 18 discontinued study treatment prematurely mainly due to disease progression and associated side effects from treatment, which in most cases resembled those associated with gemcitabine chemotherapy. However, all patients were followed for the planned two-year period. The primary endpoints were safety and immune response. The secondary endpoint was clinical efficacy at two years. The main findings were:
■
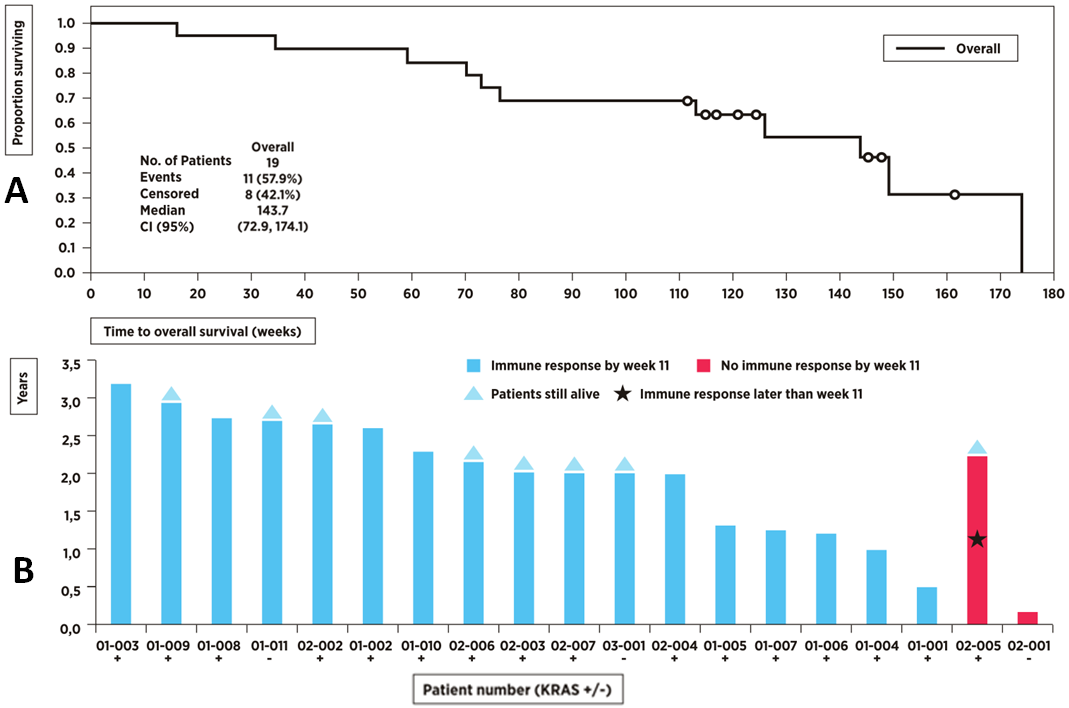
Overall survival rate at two years was 68.4% (13/19), which compares favourably with historical survival rates of 30-53% of resected pancreatic cancer patients treated with gemcitabine alone.10 The median overall survival from surgery was 33.1 months (Exhibit 8A). While there was no control arm, according to the large published ESPAC-4 study published in Q117, patients receiving gemcitabine alone live for a median of 27.6 months.
■
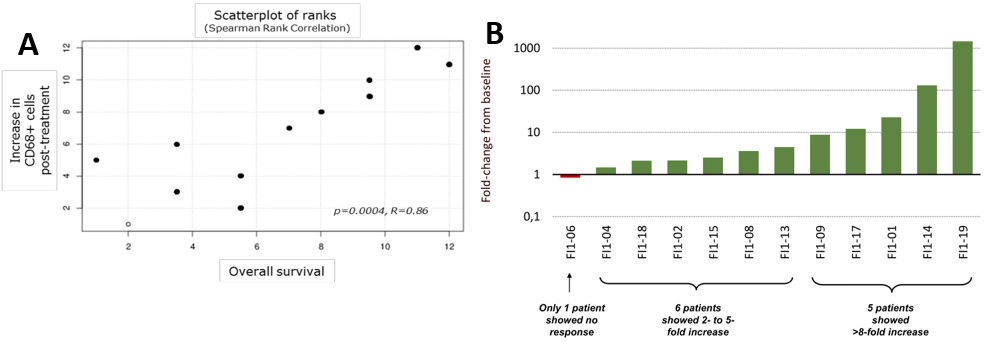
Immune response was measured using two methods: delayed type hypersensitivity (DTH) test was considered positive if TG01 injected in the skin resulted in a redness reaction of ≥5mm; T-cell proliferation assay was used to determine whether a blood sample contained TG01 specific T-cells (peripheral blood mononuclear cells). Exhibit 8B shows that 18/19 (95%) patients had immune response during the course of the study (17/18 or 89% responded early by week 11). Eight patients were still alive after the last patient completed the two-year follow up. One patient was considered a late responder with immune response detected after week 11, but still alive at the time of the analysis. The only patient without immune response died because of an unrelated cause by week 8, and therefore was not fully evaluable.
■
13 serious adverse events were reported in seven out of 19 patients. Of these three were linked to TG01 – two anaphylactic reactions and one hypersensitivity reaction, which resolved within 1-2 hours. Grade 1-2 adverse events due to TG01 included mainly expected effects such as influenza-like symptoms or injection site reactions.
The allergic reactions were seen when TG01 was administered concomitantly with chemotherapy. As a result, Targovax started a modified second cohort where TG01 is given less frequently and not during chemotherapy. This cohort of 13 patients will complete the study in H218. Recently, encouraging interim data from this cohort showed 100% survival and 85% immune activation at one year while no allergic reactions were seen.
Exhibit 8: Phase I/II TG01-01 study – survival and immune response
|
|
|
|
Overall, we find the first data set to be positive since the vaccination with TG01 was associated with 89% of the patients responding early with a production of mutant RAS specific T-cells. Only three late allergic reactions were reported, while otherwise the treatment was well tolerated. Recently released data from the modified cohort showed that there were no allergic reactions during the first year of treatment. While not a controlled trial, in the context of a very recent ESPAC-4 study, the median overall survival compares well: 33.1 months (TG01-01 study) versus 27.6 months (ESPAC-4 study). TG01-01 study also echoes the early Norsk Hydro trials, which supports further investigation of TG01.
TG01 is being tested in pancreatic cancer in Phase I/II trial. Pancreatic cancer, more precisely pancreatic adenocarcinoma, is responsible for 7% of all cancer-related deaths – the fourth leading cause of cancer deaths and 11th most common cancer diagnosed in the US. Pancreatic cancer is somewhat unique among cancers in that there has been very little progress over the past four decades in prolonging survival rates when compared to other types of cancer. This challenge has been compounded by the facts that the disease is usually diagnosed in a late stage; even if the tumour is resectable, the surgery is complicated and dangerous with high recurrence rates; and the tumour is relatively resistant to chemotherapy. This has meant that the overall five-year survival is still around 8%.
An estimated 54k new cases will be diagnosed in the US in 2017.16 Targovax’s current pancreatic cancer trial includes patients with stage I or II disease, who underwent resection surgery. Around 20% of cases are stage I or II at diagnosis (Cancer Research UK); we therefore calculate an addressable population of c 24,000. Surgery is the primary mode of treatment; however, because around half of patients are diagnosed with a distant disease, often the resection is palliative.
For resected pancreatic cancer, there is no approved treatment, implying a high unmet need. For non-resectable cancer chemotherapy options include gemcitabine alone or in combinations with several other agents such as capecitabine, erlotinib (Tarceva, Roche) and nab-paclitaxel (Abraxane, Celgene). The non-gemcitabine regimen FOLFIRINOX has been shown to be somewhat more effective than gemcitabine alone in a certain subset of patients. Recently, the standard of care chemotherapy (adjuvant) in resected disease has been changed to gemcitabine plus capecitabine (GemCap).
Tarceva brought in $229m in sales for Roche in 2016, second highest to Abraxane with $367m. Both drugs accounted for the majority of the pancreatic cancer market, which was $760m in 2016 (EvaluatePharma). With Tarceva’s patent expiring over the next couple of years, sales are forecasted to decrease.
TG02 is also being tested in colorectal cancer patients in Phase Ib trial. Colorectal cancer is the most common type of gastrointestinal malignancy and the third most common cancer overall. The five-year survival rate of colorectal cancer patients is 65% in the US, with rates being as high as 95% for stage I cancer at diagnosis, falling to 10% for stage IV patients. The majority of cases can be prevented and if detected early, could be cured. Screening programmes are therefore a major factor in the decline of colorectal cancer incidence. Survival rates were further substantially improved over the last decade due to advances in systemic therapy. Before mid-1990, the only approved chemotherapeutic drug was 5-fluorouracil, while currently the therapeutic treatment landscape is rather fragmented with established chemotherapeutic agents, but also a number of innovative biological drugs (bevacizumab [Avastin], cetuximab [Erbitux], nivolumab [Opdivo], pembrolizumab [Keytruda], regorafenib [Stivarga]) (emedicine.com). Avastin, humanised monoclonal antibody to VEGF, was the first anti-angiogenesis drug approved in combination with chemotherapy, which improved overall survival. The drug brought $3.9bn in sales for Roche in this indication in 2016, the bulk of the total market size of $7.1bn (EvaluatePharma). Although patents are to expire in 2019, the consensus estimates it will still earn $2.6bn with size staying flat at $7.1bn by 2022.
Keytruda and Opdivo are the only checkpoint inhibitors approved for patients with unresectable or metastatic colon cancer that are identified as having a biomarker referred to as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) (Keytruda is approved for any solid tumour with MSI-H or sMMR despite location). The MSI-H or dMMR constitutes only 5% of metastatic colorectal cancer patients and 60-70% of patients still did not respond to these CPIs. The consensus estimates that Keytruda’s sales will reach $10bn by 2022 with main indications being non-small cell lung cancer and melanoma, while sales for colorectal cancer will be only c $140m at that time (EvaluatePharma). Opdivo received the approval from the FDA in August 2017. Roche’s Tecentriq (atezolizumab, anti-PD-L1) is in Phase III for this indication.
The American Cancer Society estimates c 96,000 new cases of colon cancer with be diagnosed in the US in 2017. Targovax’s current trial includes patients with locally recurrent rectal cancer and it has indicated that the next trial could be in unresectable advanced colorectal cancer with standard of care chemotherapy or checkpoint inhibitor. According to Cancer Research UK, around one-quarter of patients have metastasis at diagnosis; we therefore calculate that the addressable patient population is c 53,000.