Paxalisib was licensed from Genentech in 2016. The deal included a US$5m upfront payment and a US$1m milestone payable with the first commercial sales, as well as undisclosed royalties. Concurrent with the licensing deal, Kazia also acquired the privately held company, Glioblast, to support the development of the drug. The Glioblast transaction also carries two milestones payable to the prior Glioblast shareholders: A$1.25m at completion of Phase II and an undisclosed milestone valued at A$3.4–4.2m. The drug is currently protected by patents (US8883799B2 among others) until 2032 in the US.
The drug is being developed for GBM, the most common, malignant primary brain cancer and the most common brain cancer in adults. Approximately 11,500 patients are diagnosed with GBM each year in the US. GBM tumors are characterized by invasive and diffuse growth, which makes complete surgical removal difficult. Standard treatment for GBM entails surgical resection of the tumor followed by radiotherapy and concurrent chemotherapy with temozolomide (also known as TMZ, marketed as Temodar and generics), followed by adjuvant chemotherapy with the same drug to treat the residual infiltrative component of the tumor. Despite this aggressive treatment, the disease invariably returns, resulting in a five-year survival rate of only 5%.
GBM has also been a difficult disease to develop drugs for. The only drug approved to date for first line disease is TMZ. Additionally, the medical device Optune (from Novocure) and the carmustine (a chemotherapy drug) eluting wafer Gliadel (Arbor Pharmaceuticals) are approved for the treatment of first-line GBM. Bevacizumab (Avastin and biosimilars) is approved in the US for recurring disease, but is not approved in Europe and other geographies for this indication. There has been some interest in developing checkpoint inhibitors such as drugs targeting PD-1 for the treatment of GBM, but these efforts were recently stymied by the recent failure of Opdivo (nivolumab, Bristol-Myers Squibb) in Phase III for recurrent GBM (CheckMate 143). The negative results can be explained by the immune-privileged nature of the brain, which may prevent effective utilization of immunotherapy for this indication. Opdivo is in a separate ongoing Phase III in combination with TMZ and radiation.
The multi-kinase inhibitor Stivarga (regorafenib, Bayer) has been included in the AGILE Phase II/III study (more information below) and previously showed positive results in Phase II. This drug has anti-VEGF activity similar to bevacizumab, among a range of other kinase targets associated with neogenesis.
Another promising drug is marizomib (Bristol-Myers Squibb), a proteasome inhibitor that showed a 45% overall response rate (CR+PR) in a Phase I dosing study. The drug’s lead program is for DIPG, but it is currently being investigated for GBM in a Phase III study sponsored by the European Organisation for Research and Treatment of Cancer (EORTC).
Exhibit 2: Selection of late-stage GBM clinical programs
Drug |
Company |
Phase |
Class |
Notes |
Opdivo (nivolumab) |
Bristol-Myers Squibb |
Phase III |
PD-1 inhibitor |
Separate Phase III study recently failed |
Trans Sodium Crocetinate |
Diffusion |
Phase III |
Oxygen diffusion enhancer |
Failed to show PFS improvement in Phase II |
Enzastaurin |
Denovo |
Phase III |
PKC inhibitor |
Previously discontinued, new company hopes to enhance with patient targeting |
ADCTA |
Safe Save |
Phase III |
Dendritic cell vaccine |
Little information |
DCVax |
Northwest |
Phase III |
Dendritic cell vaccine |
In Phase III testing since 2002 |
Marizomib |
Bristol-Myers Squibb |
Phase III |
Proteasome inhibitor |
Investigator-sponsored, successful in Phase I |
Stivarga (regorafenib) |
Bayer |
Phase II/III |
Multi-kinase inhibitor |
Positive Phase II results |
Source: EvauatePharma, clincialtrials.gov, company reports.
PI3K is a key effector of the receptor tyrosine kinase (RTK) pathway and is involved in many cellular signaling processes (including those mediated by other pathways). Its relevance to oncogenesis is linked to its role in transmitting growth signals (eg from growth factor receptors such as EGFR, HER2, etc), which can become aberrantly hyperactivated in a wide range of cancers. Many characteristics of cancer such as hyperproliferation, resistance to apoptosis and metastasis can be linked to the constitutive activation of this signaling cascade. Moreover, this pathway is implicated in both solid and hematologic tumor types.
Exhibit 3: PI3K signaling pathway
|

|
Source: Edison Investment Research
|
PI3K inhibitors as a class have seen some of the most intense development activity in oncology of any group of molecules. There have been a number of drugs approved in the PI3K inhibitor class (Exhibit 4). Historically, perceptions of this drug class have been marred by concerns over safety. This class can be potent inhibitors of immune cell proliferation, which can lead to opportunistic infections, as well as a range of other potentially fatal complications. These adverse effects appear to be both features of the class and drug specific. The first approved PI3K drug, Zydelig (idelalisib), famously had six ongoing clinical trials halted in 2016 when it was revealed that it was associated with high death rates. Subsequently approved PI3K inhibitors Aliqopa (copanlisib, Bayer) and Copiktra (duvelisib, Verastem) were also associated with fatal complications.
However, the story has somewhat changed with the approval of Piqray (alpelisib, Novartis) in 2019. Unlike the other drugs in this class, Piqray is approved for the treatment of breast cancer (as opposed to hematologic cancers) and shows a much better safety and tolerability profile. This fact demonstrates that fatal complications are not an absolute class feature. It has been postulated that some of the most negative class features of these drugs are associated with inhibiting PI3Kδ. Unlike many other isoforms of PI3K, PI3Kδ is expressed predominantly in hematopoietic cells and its inhibition may be associated with opportunistic infections and inflammatory adverse events (AEs; such as colitis and pneumonitis), as well as being essential for efficacy in hematologic cancers. Indeed, Piqray, which does not target this isoform, is better tolerated. However, PI3Kδ inhibition alone cannot account for the variation in safety profiles seen with these drugs. Aliqopa is a pan-PI3K inhibitor and thus inhibits PI3Kδ as well and has indeed been associated with a risk of fatal infection, but has much lower rates of the inflammatory complications more frequently associated with Zydelig and Copiktra.
Exhibit 4: Approved PI3K inhibitors
Drug |
Company |
Approved |
Indication |
Isoforms |
Safety |
Zydelig (idelalisib) |
Gilead |
2014 |
CLL, FL, SLL |
δ |
Black box for fatal infections, hepatotoxicity, and GI complications |
Aliqopa (copanlisib) |
Bayer |
2017 |
FL |
α,β,γ,δ |
Warnings for fatal infections, hyperglycemia, and cytopenias |
Copiktra (duvelisib) |
Verastem |
2018 |
CLL, FL |
γ,δ |
Black box for fatal infections and fatal GI, skin and lung complications |
Piqray (alpelisib) |
Novartis |
2019 |
HR+ HER2- breast cancer |
α |
Generally more tolerable, but with high rates of diarrhea, rash and hyperglycemia |
Source: Drug labels. Note: CLL=chronic lymphocytic leukemia, FL= follicular lymphoma, SLL=small lymphocytic leukemia.
The safety issues with this class of drug have contributed to some of the difficulties in developing new molecules. There are a large number of discontinued programs, including many from major drug developers. According to EvaluatePharma, approximately 50 such programs have been discontinued after entering the clinic. It should be noted that neither Kazia nor Genentech has reported any major safety issues with paxalisib to date (more information below). However, due to the history of this class, safety is likely to remain at the forefront of investors’ minds when evaluating the viability of the project.
Paxalisib is not the first PI3K inhibitor that has targeted GBM. Buparlisib (BKM120) developed by Novartis was examined in a Phase II GBM study but failed to show efficacy. The drug had off-target effects on tubulin, which limited its dosing. The focus of the drug was later shifted to other solid tumors, and it was subsequently divested (to Adlai Nortye) in 2018. Another PI3K inhibitor that has been examined in GBM is PX-866 (Cascadian), which entered Phase II but failed to meet its primary endpoints and was discontinued. Voxtalisib (Exelixis/Sanofi) was tested in Phase I studies against high-grade glioma, but not pursued further for this indication and later abandoned. Finally, AZD-8055 (AstraZeneca) was tested in patients with recurrent glioma in a Phase I investigator-sponsored trial, but there was no further follow-up. The above list is not exhaustive and there may be other PI3K drug candidates that were not successful in GBM.
The design and clinical history of paxalisib
The main differentiating factor between paxalisib and most other PI3K inhibitors is that it can penetrate the BBB and therefore has the potential to treat cancer in the brain. The BBB is not a discrete structure but rather the network of cardiovascular endothelium that lines the blood vessels of the brain, which control what type of molecules can exit the blood and enter the mass of brain tissue. The BBB is effective at preventing a wide range of toxins, drugs and other small molecules from entering the brain. More lipophilic small molecules can diffuse across the cell membrane of endothelial cells, but many of these are scavenged by P-glycoprotein (PGP or multidrug resistance protein). The protein binds a wide range of drug-like molecules and actively transports them out of BBB endothelial cells. Interpersonal variation in the expression levels of this protein is a major factor determining how susceptible different people are to treatment with central nervous system (CNS) targeted drugs. PGP is one of several efflux transporters that ferry molecules out of the brain, each of which recognizes different sets of structural motifs on small molecules and prevents these classes from crossing the barrier. The design of drugs to target the CNS involves generating a sufficiently lipophilic molecule that does not interact strongly with any of these classes of efflux transporters. Paxalisib was specifically designed with these factors in mind in order to achieve high concentrations in the central nervous system. Paxalisib was shown in preclinical studies to cross the BBB freely and demonstrate a pharmacodynamic effect in normal mouse brain tissue, as well as efficacy in an intracranial mouse GBM tumor model.
Paxalisib is a potent pan-PI3K inhibitor, meaning it inhibits all the isoforms of class I PI3K enzymes α,β,γ,δ. Additionally, the drug was engineered to inhibit the protein mTOR, an additional oncogene in the same pathway as PI3K and with overlapping relevance to tumor neogenesis (see Exhibit 3). Dual PI3K/mTOR inhibitors could be considered a class of drug in their own right, as many such molecules have been developed, including gedatolisib from Pfizer (and potentially others), which is currently in Phase II for breast cancer.
The drug was initially tested in the clinic by Genentech in a Phase I of patients with advanced glioma. The dose-ranging study examined doses of drug from 2mg to 65gm and found a maximum tolerated dose (MTD) at 45mg. The dose-limiting toxicity (DLT) seen in the 65mg arm was two cases of grade 3 mucosal inflammation. This AE was also seen in a significant portion of patients in the 45mg arm (4/8 patients, one at grade 3), although it was not dose limiting. Other common AE were fatigue (30% of all patients), hyperglycemia (28%), nausea (23%) and rash (17%), but these were all predominantly mild to moderate. This AE profile is consistent with the drug class but, importantly, is not characterized by signs of the potentially fatal adverse effects seen in some other examples, such as opportunistic infections or severe GI or liver toxicity.
The efficacy results for all the patients on the study are presented in Exhibit 5. A dose-response relationship is apparent, with increasing rates of tumor inhibition at higher concentrations. The best responses (based on RANO response assessment criteria) were 3/6 with stable disease in the 45mg arm and 5/6 in the 65mg arm. Although none of the patients on the study achieved a partial response (PR, defined as a 50% reduction in tumor burden) or better, the dose-response correlation observed is indicative of clinical activity.
Although Genentech divested the drug shortly after performing this study, there are several reasons to consider the molecule worthy of further investigation. First, the patient population in this study was heavily pre-treated, with patients having a median of three prior therapies, and 70% of the patients included on the study were diagnosed with grade 4 disease. This is a very sick population, with survival typically measured in months. Also, glioma has historically been a notoriously difficult disease to treat, with very limited treatment options. For comparison, TMZ had 2% complete responses (CRs) and 6% PRs when tested after the first relapse, as well as 43% stable disease. These results are statistically comparable to those seen in the Genentech study after the third relapse. It is worth noting that TMZ is not approved for treatment of the disease in this setting (although there are a number of regimens for its use as a salvage treatment), but instead as an adjuvant therapy in the first line. Similarly, paxalisib might have a more pronounced impact in a less heavily treated setting as well, which is consistent with the direction that Kazia took the drug in further studies.
Exhibit 5: Dose response of GDC-0084 in Phase I
|

|
Source: Wen et al. 2015. Note: MTD was identified as 45mg (blue bars) in the study. The upper red dotted line marks the 25% increase in tumor size (sum of product diameters) that is the maximum cut-off for stable disease according to the RANO criteria.
|
Using the knowledge gained from the Genentech Phase I study, Kazia designed a Phase IIa study to address several unanswered questions. First, the company shifted gears and instead of targeting a heavily pre-treated population as in the Phase I, it opted to enroll newly diagnosed patients. The drug was recontextualized from a rescue treatment to an adjuvant treatment following initial resection, radiation treatment and treatment with TMZ. This move is somewhat contrary to the classic clinical algorithm, in which a drug is developed initially to treat the sickest patients, but is well reasoned. As cancer progresses, it often becomes both harder to treat as it becomes more heavily mutated and heterogeneous, and additionally, the patients become less resilient and intolerant to aggressive treatment. This revised approach opens the potential to both use more aggressive dosing in these new patients and to see better responses. Moreover, due to the lack of available treatments for the disease, there exists significant unmet medical need even in these newly diagnosed patients. A final change in strategy from the Genentech Phase I is that instead of enrolling patients with glioma of all types, the Phase IIa exclusively enrolled newly diagnosed GBM patients. This may provide more conclusive results by using a more homogeneous patient population and the results may be more comparable to other studies, as the majority of the research in the space has been devoted to GBM.
This study also specifically targeted the population of patients that lack methylation of the MGMT promoter (as confirmed through genomic analysis), which predicts that patients in this population are not good candidates for alkylation chemotherapies such as TMZ. Although TMZ is included in the protocol as part of the standard of care, these patients should effectively be resistant to it. Based on a 2017 meta-analysis of MGMT status in GBM aggregating 34 independent studies,1,808 (50%) of the pooled 3,598 patients whose methylation status was known were unmethylated, although there is a high degree of variability between individual studies.
The first part of the Phase IIa was a dose-ranging study (with a classic 3+3 protocol design). The goal of this portion of the study was to reinvestigate the MTD in this new patient group. The study enrolled nine patients into the dosing study and found a new higher MTD of 60mg following mucositis and hyperglycemia DLTs at 75mg. This is encouraging because the drug will be more likely to demonstrate treatment effects at this higher dosing level, which is being used in the second part (the expansion cohort) of the study. The most severe AEs in this stage were rash, mucositis and hyperglycemia, consistent with earlier results, and reinforcing the more attractive risk profile compared to other PI3K inhibitors.
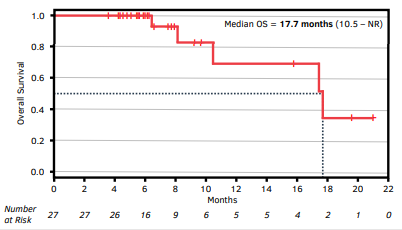
Following identification of the new MTD, Kazia proceeded to the expansion cohort (n=21), a two-arm, open-label study. The study will examine the role of any food effects in the administration of the drug by administering it in either the fed or fasted state between the two arms. This study phase is currently ongoing, but Kazia recently presented an interim analysis at the 2020 American Association for Cancer Research (AACR) virtual meeting (Exhibits 6 and 7). The company reported a median progression-free survival (PFS) of 8.5 months and an overall survival of 17.7 months for the entire study population (n=27 evaluable).
Exhibit 6: PFS from Phase II study of paxalisib
|
Exhibit 7: OS from Phase II study of paxalisib
|

|
|
|
|
|
Exhibit 6: PFS from Phase II study of paxalisib
|

|
|
|
Exhibit 7: OS from Phase II study of paxalisib
|

|
|
|
The data from this interim analysis compare favourably to historical controls in patients with unmethylated MGMT (Exhibit 8). A word of caution should always be given when drawing conclusions using historical controls as these comparisons lack statistical rigor and there may be imbalances in the patient populations. However, these data provide the clearest picture to date that the drug has the potential to provide a benefit in these patients.
Exhibit 8: Overall survival and PFS in GBM with unmethylated MGMT promoter treated with radiotherapy plus TMZ
|
Median overall
survival (months) |
Median PFS
(months) |
PFS
at 6 months |
Two-year
survival rate |
Hegi et al NEJM 2005 |
12.7 |
5.3 |
40% |
14% |
Nabors et al, Neuro-Oncology 2015 |
13.4 |
4.1 |
N/A |
N/A |
Gilbert et al, JCO, 2013 |
14.0 |
5.7 |
N/A |
N/A |
AVAGLIO ASCO, 2013 |
14.6 |
5.8 |
N/A |
N/A |
RTOG-0825, ASCO, 2013 |
14.3 |
N/A |
N/A |
N/A |
Average |
13.8 |
5.2 |
|
|
Source: Edison Investment Research; Hegi et al N Engl J Med 2005;352(10):997-1003; Nabors et al. Neuro-Oncology 2015 17(5):708-717; Gilbert et al. J Clin Oncol 2013 31(32):4085-4091. Note: RTOG = Radiation Therapy Oncology Group
Recruitment for the Phase IIa study completed in February 2020 (n=21). The study has a planned completion date in December 2020, so we expect top-line final data from the study in early 2021. The company also has a planned presentation of additional interim results at the Society for Neuro-Oncology meeting in November 2020.
In December 2019, it was announced that Kazia had been asked to submit paxalisib for inclusion in the GBM AGILE study. The AGILE study is a Phase II/III program sponsored by GCAR, a consortium of academic neurologists and neuroscientists. The study is designed to test a range of different treatments for GBM against a common control group. It is innovative in many of its features including the ability to expand its criteria to include additional new treatments, such as paxalisib. The study is currently ongoing, but Kazia is currently still preparing to include paxalisib and start enrolling patients later this year. It stated that up to 200 patients may be enrolled in the paxalisib arm. The precise protocol is a matter for discussion with the trial coordinators, but we assume that the drug will be used in the same context as the ongoing Phase II study (as an adjuvant following resection, radiation and TMZ).
Kazia plans to use the results from the AGILE study as the pivotal clinical trial data to support approval of paxalisib. Based on feedback from the FDA, it is confident that AGILE can be used to support approval. There are several risks and benefits to this approach. First, using it as the pivotal trial should substantially reduce the financial burden on the company. Kazia still has some financial obligations associated with the study, which are undisclosed, but we expect them to be smaller than hiring a contract research organization (CRO) for the same purpose. For the purposes of our valuation, we assume that the costs to the company for the AGILE study will be approximately US$22m, roughly half of what we would expect for a similar cohort otherwise. However, the trade-off is that Kazia will have substantially less control over the conduct of the trial. Although the study could potentially be used as a pivotal study, the approval of paxalisib in particular is not the expressed purpose of the trial, and it cannot be ensured that the trial’s sponsors will make decisions regarding the drug that are in the best interests of approval. Decisions could be made that would prevent using the trial as a pivotal study even if the results are positive. Moreover, Kazia does not have control over the timeline for inclusion in the study. Based on our estimates, we expect the study to take around three years to provide pivotal results for paxalisib, but it cannot be ensured that the sponsors will enroll patients into this arm at a sufficient pace to meet these timelines. That being said, we believe that inclusion in the study is a major opportunity for Kazia because we expect that otherwise it would not be able to support running a pivotal trial at its current market capitalization without a major development partner, a process that carries its own sets of risks and uncertainties.
In August 2020, it was announced that the program had received a FTD from the FDA. This status is designed to improve the pace at which drugs are approved for certain areas of significant unmet need by increasing engagement with the FDA. Kazia will be entitled to more frequent meetings and communication with the agency to discuss its clinical development plan. Additionally, the product may be evaluated in a rolling review process, in which individual portions of the NDA can be submitted to the FDA as they are produced (as opposed to submitting them all as a single package), and feedback on these portions will be provided. Both of these privileges are expected to remove some of the potential friction involved in the regulatory process. Finally, products that are selected for FTD are also eligible for accelerated approval and priority review.





















