Company description: Fund-raising enables strategy execution
4SC is a Munich-based biotech company focused on developing small-molecule drugs for cancer. Together with the successful fund-raise, the company announced earlier this year a refocused R&D strategy, which centres the development of its assets for dermatological cancers. 4SC’s lead product resminostat is in a pivotal study in CTCL, which was initiated in Q416 with the results expected in H119. Resminostat is partnered with Yakult Honsha in Japan, where most recently a Phase II study in first-line HCC and a Phase I study in biliary tract cancer have been completed. Yakult indicated that it plans to continue the development in biliary tract cancer with a Phase II study to be initiated shortly. 4SC-202 and 4SC-208 are the two other core portfolio assets in earlier stages, also for dermatological cancers, Melanoma (Phase II ready), MCC (pivotal stage after melanoma study) and BCC (preclinical), respectively. 4SC was founded in 1997, listed on the Frankfurt stock exchange in December 2005, and has 47 full-time employees.
Valuation: €344m or €11.2/share
Our updated rNPV-based valuation stands at €344m or €11.2/share, compared to €124m previously, including estimated cash of €43.4m after the fund-raising. To reflect 4SC’s updated strategy we have overhauled our model, making substantial changes. The core portfolio includes resminostat for CTCL, 4SC-202 for MCC and 4SC-208 for BCC. We assume that 4SC will develop and market the products in these indications itself. Out-licensed assets or indications include resminostat for biliary tract cancer in Japan developed and marketed by Yakult (our scenario), and two of 4SC’s other out-licensing deals that have disclosed financials (Link Health and Maruho).
Financials: Well-funded to several R&D events
Following the recent €41m gross (estimated €40m net) capital raise, we forecast cash reach into FY20 and have increased our R&D forecasts to €14.0m, €16.8m and €12.6m in 2017, 2018 and 2019, respectively. This will enable 4SC to advance its clinical programmes for resminostat, 4SC-202 and 4SC-208 significantly. We do not include potential milestones from any ongoing development of resminostat with its partner Yakult or other milestones from non-core assets. As such, if the partnerships progress, there is potential for additional injections of non-dilutive cash.
Sensitivities: Typical biotech risks apply
4SC is subject to sensitivities typical of biotech drug development, including the unpredictable nature of clinical trials, the success or failure of competitors and changing market dynamics. The company has reduced its financial risk with its recent fund-raising (estimated €40m net), enabling it to progress its lead product resminostat (ex Japan) in pivotal development for CTCL alongside earlier-stage products 4SC-202 and 4SC-208.
Core portfolio focused on dermato-oncology
4SC’s core focus is on dermato-oncology utilising discrete and potentially effective therapeutic mechanisms to target certain cancers (CTCL, MCC and BCC), which the company believes represent the fastest opportunities to reach the market. For the time being, assets or indications that are beyond dermato-oncology are being considered for out-licensing. Notably, the company’s assets are well suited for use in various combinations, especially with cancer immunotherapies.
Exhibit 3: Clinical development focus – ongoing and planned studies
|

|
|
|
Resminostat – lead asset, HDAC inhibitor
Resminostat is an orally administered histone deacetylase (HDAC) inhibitor with potential to act as a monotherapy and/or in combination therapy with other anti-cancer drugs. Resminostat inhibits HDAC classes I, IIb and IV, but has alongside pronounced activity against HDAC 6, which could potentially facilitate inhibition of tumour growth, cause regression and bolster the innate immune response. In short, it has the ability to re-programme cancer cells.
Resminostat has shown encouraging anti-tumour activity with good tolerability in patients with advanced cancers in Phase I and Phase II studies. It has been tested in patients in a range of blood and solid tumours, both as a monotherapy and in combination with chemotherapies in the EU and Asia. It was generally well tolerated, with adverse effects in line with or better than other HDACs, principally GI effects (nausea and diarrhoea), fatigue and thrombocytopenia. Importantly, there were no severe liver, cardiovascular or GI bleeding side effects. Exhibit 4 below outlines the body of data (>300 patients to date) that has been built around Resminostat in Phase I and II studies.
Exhibit 4: Overview of completed resminostat clinical studies
Study |
Study phase |
Number of patients |
Study design |
Publication |
Resminostat in combination with sorafenib in advanced hepatocellular carcinoma (HCC) |
II |
170 |
A multinational, multicentre, randomised, double-blind, placebo-controlled Phase II study to evaluate resminostat in combination with sorafenib for the first-line treatment of advanced HCC, conducted by partner Yakult Honsha. |
Resminostat and sorafenib combination therapy for advanced hepatocellular carcinoma in patients previously untreated with systemic chemotherapy |
4SC’s resminostat in combination with S-1 chemotherapy in pre-treated biliary tract cancer. |
I |
27 |
A multicentre, open-label Phase I study of 4SC’s resminostat in combination with S-1 chemotherapy in 27 Japanese patients with pre-treated biliary tract or pancreatic cancer. |
Presented at the European Society for Medical Oncology (ESMO) conference in September 2017 |
SHELTER – Combination of sorafenib and resminostat in hepatocellular carcinoma (HCC) |
II |
57 |
A multinational, multicentre, single-arm Phase II study to evaluate efficacy, safety and pharmacokinetics of resminostat and the treatment combination of sorafenib and resminostat in patients with HCC exhibiting progressive disease (PD) under sorafenib treatment. |
Resminostat plus sorafenib as second-line therapy of advanced hepatocellular carcinoma – The SHELTER study |
SAPHIRE – Resminostat in Hodgkin’s LYMPHOMA |
II |
37 |
A multinational, multicentre, single-arm Phase II study to evaluate the efficacy, safety and pharmacokinetics of resminostat in patients with relapsed or refractory HL. |
|
NSCLC |
I/II |
9/108 |
Dose escalation in patients with advanced, metastatic, or recurrent NSCLC who have previously received one platinum-based chemotherapy treatment. The resminostat/docetaxel combination proved to be safe and well tolerated in all dose levels tested. |
Phase I/II study of docetaxel combined with resminostat, an oral hydroxamic acid HDAC inhibitor, for advanced non-small cell lung cancer in patients previously treated with platinum-based chemotherapy. |
SHORE – Combination of Resminostat and FOLFIRI in Colorectal Cancer (CRC) |
I/II |
17 |
A national, multicentre Phase I/II study to evaluate safety, tolerability, pharmacokinetics and efficacy of resminostat in combination with a second-line treatment in patients with K-ras mutated advanced CRC. The Phase II part of the study was not conducted. |
|
Resminostat in advanced solid tumours |
I |
12 |
A mono-centre, single-arm Phase I study of resminostat in Japanese patients with solid tumours, conducted by Yakult Honsha. |
A phase I study of resminostat in Japanese patients with advanced solid tumours. |
First-in-human study of resminostat in advanced solid tumours |
I |
19 |
A first-in-human, mono-centre, single-arm, open-label, dose-escalation study of resminostat in patients with advanced solid tumours. |
First-in-human, pharmacokinetic and pharmacodynamic phase I study of Resminostat, an oral histone deacetylase inhibitor, in patients with advanced solid tumours |
Source: 4SC and clinical trials.gov
CTCL – resminostat could be the first HDACi approved for CTCL in Europe
Currently 4SC is running a pivotal trial (the RESMAIN study) with resminostat to evaluate it for maintenance treatment in patients with advanced-stage CTCL who have achieved disease control with prior systemic therapy. Exhibit 5 summarises the details. Resminostat is focused on patients with late stage, incurable disease. Such patients typically receive many different lines of therapy over their lifetime as none of the current therapeutic options achieve stable disease for long periods, with virtually all patients progressing after three to four months on average. Should resminostat show efficacy as a maintenance therapy – prolonging the period patients are stable and not progressing – it means that the target population for resminostat is virtually all late stage patients who have received at least one line of therapy.
Overall 10-year survival rates vary from 98% if diagnosed at stage IA to 20% if diagnosed at clinical stage IVB. Theoretically, this means that patients could receive multiple cycles of therapy-resminostat-therapy-resminostat and so on. Because of the unique positioning as a maintenance therapy, the company believes this would clearly differentiate resminostat against two other HDAC inhibitors approved in the US ,vorinostat (Zolinza, Merck & Co) and romidepsin (Istodax, Celgene), which are indicated for patients with progressive disease in second or later lines and are typically used only once as a single line of therapy. Notably, no HDAC inhibitors have been approved for this indication in Europe.
Top-line results for resminostat in CTCL are expected in H119, and if they are positive, 4SC has indicated that it would submit the data as soon as possible to the regulatory authorities for market approval. The company expects efficacy data from the ongoing RESMAIN study in Europe to be sufficient for the FDA as well, but there might be smaller and shorter bridging safety studies required to file for the approval in the US. This could be resolved while the RESMAIN study is ongoing and 4SC expects to submit resminostat for market approval in the US and Europe at the same time. If approved, resminostat would be the first HDAC inhibitor approved for CTCL in Europe and the first and only therapy approved for maintenance in the US and Europe. The company is targeting orphan drug designation in CTCL in Europe and the US, which would provide 10 years’ market exclusivity in Europe and seven years in the US.
Exhibit 5: Overview of the RESMAIN clinical study
Aim |
To determine whether resminostat will be able to delay worsening of disease in patients with advanced stage mycosis fungoides (the most common form of CTCL) or Sézary syndrome (about 5% of all cases of CTCL) who have recently achieved disease control with previous systemic therapy. |
Summary design |
Multicentre, double-blind, randomised, placebo-controlled Phase II trial |
Design details |
150 patients, two-arm study. Active arm: 3 tablets, 5 days’ treatment followed by 9 days’ rest (cycles until progress or unacceptable to toxicity). |
Inclusion criteria |
Patients with histologically confirmed mycosis fungoides (Stage IIB-IVB) or Sézary syndrome with a complete response, partial response or stable disease after at least one prior systemic therapy according to local standards, adequate haematological, hepatic and renal function |
Exclusion criteria |
Patients with progressive disease, elongated baseline corrected QT interval, concurrent use of any other specific anti-tumour therapy |
Primary endpoint |
Progression-free survival (PFS) |
Secondary endpoints |
Time to symptom worsening (TTSW), specifically pruritus |
Start date |
End 2016 |
Completion dates |
Final data collection early 2019 |
Source: clinicaltrials.gov
HCC and biliary tract cancer in Japan – in the hands of Yakult
As summarised in Exhibit 4, resminostat has shown efficacy and safety in combination with sorafenib (current standard first-line medication) in a subset of patients with advanced HCC in a Phase II trial conducted by 4SC’s partner Yakult and may support continued development in this indication. One possible option, although not confirmed yet, is a pivotal study with a subgroup of HCC patients who have higher baseline platelet levels. Yakult’s Phase II HCC study showed a 40% reduction in risk of death for those with higher baseline platelet levels. The company has indicated that it sees the future of resminostat in HCC in China and is actively looking to partner the drug in this, the largest market for HCC globally. We also note that 4SC has orphan drug designation in the EU and US for resminostat use in HCC with patent protection until late 2026 in Europe and until mid-2027 in the US. Therefore, the company is able to partner the development of resminostat outside Japan for non-core indications as well.
The most recent data from Yakult is from a Phase I trial with patients with biliary tract cancer, which was presented at the ESMO conference in September 2017. Resminostat was given in combination with S-1 chemotherapy to 27 Japanese patients with pre-treated biliary tract or pancreatic cancer (Exhibit 6). While primarily a safety (well-tolerated regimen identified) and dose finding study, secondary endpoints provided early insights in potential efficacy:
■
Tumour shrinkage/disease stabilisation has been observed in all patients with biliary tract cancer.
■
Median overall survival (OS) was 10.2 months, while median progression free survival (PFS) was 5.5 months in biliary tract cancer patients.
Due to low patient numbers, any evaluation of efficacy should be cautious; however, reported historical OS and PFS in biliary cancer patients after second-line treatment have been very poor at 6.6 months (OS) and 3.0 months (PFS) with a very low response rate (3.4%). Following the publication of the new data, Yakult indicated that it is planning to conduct a Phase II trial in the near future in biliary tract cancer.
Exhibit 6: Design of the Phase I trial with biliary tract cancer patients (Yakult)
Aim |
To evaluate resminostat monotherapy and S-1/resminostat combination therapy in biliary tract or pancreatic cancer |
Design details |
Phase I, open-label, dose-escalation study to evaluate the dose-limiting toxicities of resminostat monotherapy and S-1/resminostat combination therapy in Japanese patients with unresectable/recurrent biliary tract or pancreatic cancer to determine the recommended dose regimen/s to be used in subsequent Phase II trials conducted by partner Yakult Honsha. |
Primary endpoint |
Dose-limiting toxicities of resminostat monotherapy and S-1/resminostat combination therapy. |
Secondary endpoints |
Assess safety and pharmacokinetics, assess efficacy endpoints – overall response, progression free survival, overall survival. |
4SC-202 – promising combination partner for checkpoint inhibitors, HDAC inhibitor
4SC-202 is 4SC’s second epigenetic drug; that selectively inhibits HDAC isoforms 1, 2 and 3 – for more details regarding its mechanism of action please see 4SC’s website. 4SC-202 is once daily and orally administered.
A relatively recent focus in solid tumour indications is the use of checkpoint inhibitors (CPI); however, there remain a majority of patients who neither respond to therapy nor experience durable responses. Data presented by 4SC indicate that there is significant potential for 4SC-202 in combination with anti-PD-L-1 antibodies to expand the treatable patient population. Exhibits 7 and 8 below outline data where 4SC-202 was used alone and appear to indicate it reduces the tumour burden as a monotherapy more effectively than anti-PD-L1 alone and that this anti-tumour effect was also improved when used in combination with anti-PD-L1 therapy in a murine colon carcinoma model.
Exhibit 7: 4SC-202 synergises with anti-PD-L1 therapy – tumour size
|
Exhibit 8: 4SC-202 synergises with anti-PD-L1 therapy – tumour volume
|

|

|
|
|
|
Exhibit 7: 4SC-202 synergises with anti-PD-L1 therapy – tumour size
|

|
|
|
Exhibit 8: 4SC-202 synergises with anti-PD-L1 therapy – tumour volume
|

|
|
|
4SC-202 has completed a Phase I study (see Exhibit 9 for an overview), where most importantly it proved to be safe and well tolerated by patients. In particular, the compound demonstrated promising indications of anti-tumour efficacy, both in terms of long-term stabilisation of the disease and in shrinking the actual tumour itself. Alongside this there is preclinical evidence that 4SC-202 strengthens the endogenous immune response to cancer cells.
Exhibit 9: Overview of completed Phase I study 4SC-202
Study title |
Stage |
N |
Study design |
Publication |
TOPAS – First-in-human study of 4SC-202 in hematologic malignancies |
Phase I |
24 |
The TOPAS study was a first-in-human, multicentre, single-arm, open-label, dose-escalation study of 4SC-202 in patients with advanced hematologic malignancies. |
For academic publications click here |
Source: 4SC and clinicaltrials.gov
4SC-202 next steps: Two Phase II trials and a pivotal study
4SC intends to initiate two Phase II studies of 4SC-202: firstly, in Q417 in combination with checkpoint inhibitors in patients with advanced melanoma who are refractory to treatment with checkpoint inhibitors, followed in Q118 in an investigator-initiated study in non-responding patients with microsatellite-stable gastrointestinal cancers. The company expects that the two studies are a sensible strategy because the accumulated data will allow it to assess the safety profile of 4SC-202 in combination with both anti-PD-1 and anti-PD-L1 checkpoint inhibitors and in the two patient populations with the greatest medical need; relapsed and non-responding patients. This data set would form the basis for potential partnering discussions for indications outside 4SC’s core focus and establish the recommended dose for a subsequent pivotal study in MCC, an orphan indication. If the data are sufficiently positive, 4SC-202 could be the first drug to market in a checkpoint inhibition refractory MCC population, which could prove a significant commercial opportunity.
MCC is a rare, aggressive form of skin cancer. Around 1,600 people in the US are diagnosed with MCC every year. If diagnosed early, surgical treatment is an option, but overall, approximately half of all cases will relapse. Avelumab (Bavencio, anti-PD-L1, Pfizer) was the first checkpoint inhibitor (and first therapy specifically for MCC) to be approved by the FDA in March 2017. The accelerated approval was based on a relatively small clinical trial with 88 patients with metastatic MCC. 33% of patients achieved overall response (i.e. complete or partial) and the response lasted for more than 12 months in 45% of responding patients, meaning that the majority of the patients ultimately progress after checkpoint inhibition or do not respond to CPI monotherapy. Exhibit 10 outlines the three planned studies and timelines. We note there is patent protection until late 2029 in Europe and early 2030 in the US, but MCC would qualify as an orphan disease and orphan drug designation would allow for market exclusivity for seven years in the US and ten years in Europe.
Exhibit 10: Clinical development strategy for 4SC-202
|

|
|
|
4SC-208: Downstream Hedgehog signalling pathway inhibitor
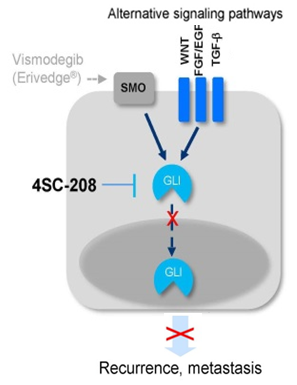
4SC-208 is a small molecule specifically targeting two kinases that are integral for Hedgehog/GLI signalling. Pre-clinical data have indicated efficacy of 4SC-208 in inhibiting this signalling pathway in a pancreatic cancer in vivo model (Exhibit 12). 4SC-208 acts further downstream to other small molecule drugs such as vismodegib (SMO inhibitor, Exhibit 11), which if demonstrated to be true could have the potential to prevent cancer stem cells from developing into tumours, causing metastases and tumour recurrence. 4SC believes that BCC could be the fastest route to market with 4SC-208 and a good first target indication for 4SC-208 since dysregulation of the Hedgehog/GLI signalling pathway is very prevalent in this disease.
BCC is the most common form of skin cancer with incidence of around 2.8m cases a year in the US alone. It rarely metastasizes (less than 0.1%) and in the majority of cases can be well managed with a range of therapeutic options, including, for example, surgical treatment. Significant problems arise if BCC is diagnosed late, which is when it causes significant morbidity and cosmetic disfigurement. Vismodegib (Erivedge, Roche) was the first Hedgehog signalling pathway targeting drug approved in 2012 for BCC, while sonidegib (Odomzo, Novartis) followed in July 2015. Both are so called SMO inhibitors indicated for recurrent locally advanced BCC in patients who cannot be treated with surgery or radiation. Notably, such advanced cases with no curative options are rare, estimated within a range of 1-10%. Patients also typically relapse after the treatment with vismodegib or sonidegib.
4SC plans to position 4SC-208 for patients who have relapsed after treatment with SMO inhibitors, but since it acts further downstream (to SMO inhibitors), it will theoretically prevent the cancer from recurring by utilising alternative GLI activating pathways (Exhibit 11). A first-line positioning in advanced BCC is also possible, depending on the pivotal trial data.
4SC has indicated that it intends to complete formal development of 4SC-208 and start a Phase I/II clinical trial early in 2019, with an early potential outline of the study shown in Exhibit 13.
Exhibit 11: 4SC-208 mechanism of action
|
Exhibit 12: 4SC-208 efficacy data
|
|

|
Source: 4SC presentation. Note: SMO – smoothened receptor.
|
|
Exhibit 13: Clinical development strategy for 4SC-208
|

|
|
|






















